Current VISN22 MIRECC Research Projects
- Cognition
- Motivation
- Social Cognition & Social Neuroscience
- Service Use and Contextual Factors
- Education Research Programs
Clinical Research Programs
The MIRECC clinical research programs address domains that affect the ability of Veterans with serious mental illness (SMI) to function in their daily lives. These include meeting their educational and occupational goals and enriching their social and family lives. The MIRECC scientific programs also address the contextual issues that affect functioning, including access to health care, access to housing, racial disparities, and poverty. Figure 1 is a graphic representation of the factors we study that affect functioning in Veterans with SMI. In the following sections, we describe our approach to understanding and treating each factor.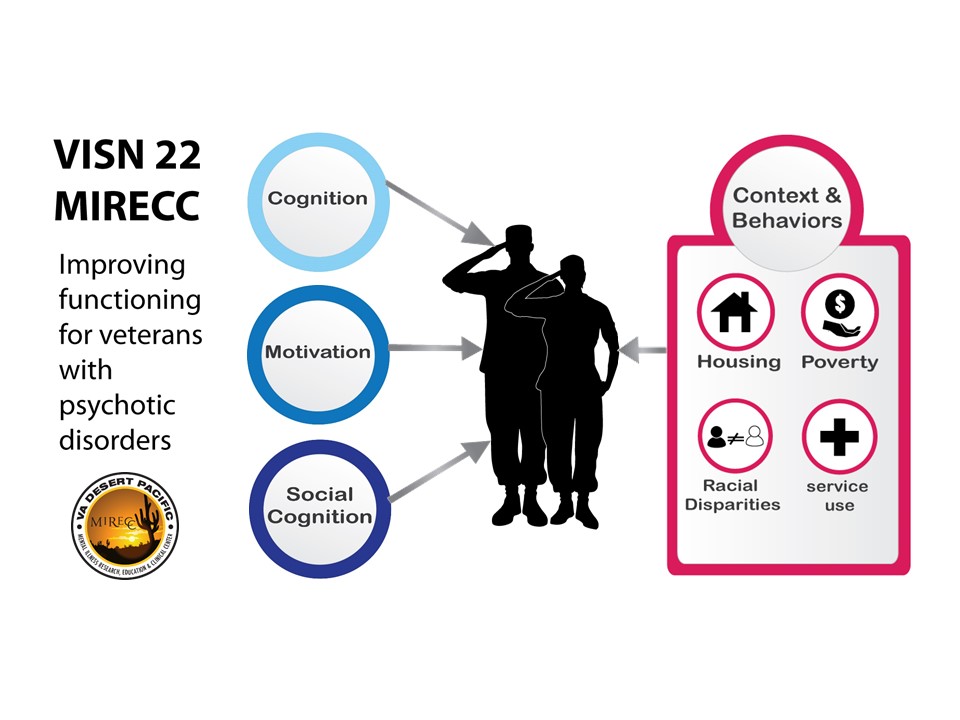
Cognition
The launch of the VISN 22 MIRECC was grounded in the pioneering work on cognition in psychosis from its members. Cognition refers to higher-level mental functions such as perception, memory, attention/vigilance, reasoning, problem solving, and speed of processing. Drs. Marder and Green demonstrated the importance of cognition for daily functioning in people with schizophrenia and headed the NIMH-MATRICS Initiatives to standardize the design of clinical trials in schizophrenia. At the same time, Drs. Braff, Geyer, and Light were conducting seminal cross-species work on the neurophysiological basis of the contributions to cognitive and functional disability in schizophrenia. Our MIRECC investigators have received many grants over the years to support projects to deconstruct complex cognitive processes into their basic elements and to understand how these core deficits contribute to daily functioning for Veterans with psychotic disorders. For example, we have shown that deficits in early visual and early auditory perception both relate well to community integration.
As a complement to the focus on cognition as a predictor of functional outcome, MIRECC investigators have been closely involved with development and validation of interventions for cognitive impairments. Drs. Marder and Green led the NIMH MATRICS Initiative which has stimulated research on innovative drugs to improve both cognition and negative symptoms in individuals with schizophrenia. Studies of promising novel agents for improving cognition are ongoing in the MIRECC, as are studies of neuroplasticity-based cognitive remediation. For example, a study by Carol Jahshan contrasted two different cognitive therapy approaches. In related work, Drs. Braff and Light led a multi-site effort (The Consortium on the Genetics of Schizophrenia) to identify the genomic basis of neurophysiologic biomarkers and cognitive deficits in schizophrenia. Cognitive therapy is a validated therapy for cognitive impairment, and Dr. Niv has developed an informational handout on this approach for clinicians.
A promising extension of cognitive therapy involves a series of projects by Light and colleagues that examine the neurophysiological predictors of response to an auditory-based targeted cognitive training (TCT) intervention. These recent TCT studies of patients with treatment refractory schizophrenia found that the intervention improves engagement in other important psychosocial groups and activities, significantly reduces auditory hallucinations, improves verbal learning, and even improves the discriminability of speech in noisy backgrounds relevant for social functioning. Importantly, a series of studies, all led by MIRECC fellows, demonstrated that EEG biomarkers developed within our MIRECC measured at the initiation of TCT treatment robustly predicted clinical and cognitive benefits after three months of treatment, supporting biomarker-informed assignment to this effective intervention. This work was recognized as a “Top Discovery of 2018” by the Brain and Behavior Research Foundation. Drs. Joshi and Molina – both former MIRECC physician fellows – now have VA Career Development Awards to expand this work in optimizing cognitive therapy for Veterans. Light and colleagues are further extending this program of research via two recently funded NIH grants that aim to determine whether specific medications can potentiate or accelerate the benefits of TCT and to confirm that benefits are predicted by biomarkers measured at the initiation of treatment.
Following the roadmap of the NIMH MATRICS initiative and the experience with the COGS, Dr. Light contributed to leadership groups formed by the Foundation of NIH to further standardize EEG-based biomarkers for valid use in multi-site clinical trial studies. These efforts contributed to the formation of the Accelerating Medicines Program for Schizophrenia (AMP SCZ), a public-private partnership between the NIH, the U.S. Food and Drug Administration (FDA), the European Medicines Agency, and multiple public and private organizations. The overall aim of the AMP SCZ initiative is to generate biomarker tools that will considerably improve success in developing early-stage interventions for patients who are at risk of developing schizophrenia. Light co-directs the EEG core for AMP SCZ with Dr. Dan Mathalon (San Francisco VA).
Motivation
Motivation is the intensity and persistence of goal-directed behavior and includes cognitive (beliefs and attitudes), emotional (drive or volition, pleasure), and behavioral (putting forth effort), components. Motivational negative symptoms significantly interfere with daily functioning and contribute to poor community outcomes among Veterans with SMI. At the inception of the VISN 22 MIRECC, negative symptoms were poorly understood with few treatment options. Work from our MIRECC has contributed to a better understanding of negative symptoms, specifically amotivation. We participated in efforts to develop clinical rating scales and have developed parallel behavioral tasks for both humans and animals to define component processes of motivation and underlying brain mechanisms of amotivation, helping to develop efficacious interventions for Veterans.
MIRECC investigators participated in defining the core components of negative symptoms and in development of clinical rating scales. As we considered measurement of these components, it became clear that animal researchers had several effective tools adaptable for humans, resulting in our validation of objective, performance-based measures of motivation for people with psychotic disorders. These studies reveal that people with schizophrenia have deficits in the “wanting” but not “liking” components of motivation and that effort valuation contributes to overall cognitive performance. An ongoing study by Dr. Wynn is using behavioral and fMRI methods to examine among Veterans with schizophrenia four motivation sub-components critical for translating information about rewards into adaptive community functioning. These components are reward receipt, reward anticipation, effort valuation, and more complex goal-directed action selection. This study also includes Veterans with bipolar disorder, another serious mental illness with an opposing pattern of motivation: whereas schizophrenia is associated with hypo-reactivity in certain aspects of reward processing, bipolar disorder is associated with hyper-reactivity to rewards, consistent with objective measures we developed. Other work from MIRECC investigators revealed an EEG biomarker of motivation in this objective measurement, with reduced parietal alpha occurring as healthy people and rodents choose to ‘give-up.’ Our work, therefore, helps specify brain-based, reward-processing impairments that contribute to motivational disturbances and guides both clinical and preclinical studies determining such mechanisms. In our view, understanding the brain-based impairments is a vital component for developing new treatments.
Building on our translational animal and human studies, MIRECC investigators also pursued innovative psychosocial interventions for motivation. These efforts initially used techniques from social skills training to improve motivation in Veterans with psychotic disorders. Beyond psychosocial interventions, our group has been involved with establishing guidelines for clinical trials of new medications to enhance motivation. A study by Dr. Reddy evaluated a promising psychosocial treatment for motivational negative symptoms and examined a potential neurophysiological biomarker of motivation. This randomized controlled trial assessed the feasibility of a treatment for motivational negative symptoms with a novel combination of motivational enhancement techniques augmented with cognitive-behavioral therapy (MI-CBT). Veteran participants in the treatment condition, compared to the active control, showed significant and durable improvements in motivational negative symptoms. Community functioning improved at a trend level. The results also supported pupillometric markers of cognitive effort as sensitive indicators of clinical improvements in motivational negative symptoms. Hence, such novel psychosocial treatments can enhance motivation, and hopefully lead to better community integration.
Finally, our work is contributing to potential solutions for the substantial challenges of community integration for homeless Veterans by examining the role of motivation in homeless Veterans with SMI. In a series of studies, we found that measures of motivation predicted community integration in homeless Veterans over a 12 month follow up. In fact, measures of motivation were better predictors than were cognition measures. Thus, motivational deficits are important treatment targets for social integration in this population. Validated treatments (e.g., MI-CBT) and novel medications could be applied to unsheltered and recently-housed Veterans to improve motivation and ultimately community integration.
Social Cognition & Social Neuroscience
Social cognition refers to mental processes that underlie social interactions, such as perceiving, interpreting, managing, and generating responses to socially-relevant stimuli, including the emotional expressions and intentions of others. MIRECC investigators have made significant contributions to our understanding of the nature of social dysfunction in schizophrenia. Our work, as well as the work of others, indicates that individuals with schizophrenia consistently show deficits in several aspects of social cognition such as social perception, emotion processing, mentalizing, and empathy.
MIRECC investigators have taken the lead in defining the brain regions and networks that underpin social processing deficits in schizophrenia and other psychotic disorders. For example, Dr. Jimenez has shown that non-verbal social cue perception deficits in schizophrenia are associated with reduced neural activity in two key social processing brain regions (amygdala and posterior superior temporal sulcus) compared to healthy controls. In addition, functional connectivity between the posterior temporal lobe and frontal cortex was reduced in schizophrenia. Another key process underlying social functioning is empathy – the ability to understand and share the perspectives of others. Dr. Jimenez’s ongoing study explores the brain networks underlying empathy deficits in Veterans with schizophrenia, including alterations in connections within and between affective and cognitive empathy networks.
Our MIRECC group has identified several intervening variables (including motivation) through which social cognition is related to functioning. Individuals with schizophrenia show deficits in social motivation and reward processing, which directly impacts goal-directed behavior. Studies from our MIRECC showed reduced neural sensitivity in patients with schizophrenia in key reward-processing brain regions (e.g., ventral striatum, ventromedial prefrontal cortex, anterior cingulate cortex) for social, but not nonsocial, rewards.
Our investigators have played a leading role in translating social cognition and social neuroscience findings into the development of training interventions for social cognitive deficits in people with psychotic disorders. Dr. Horan and colleagues designed a 24-session, group-based intervention to improve social cognitive skills in adults with a range of psychiatric conditions, and Dr. Kern extended social cognitive training to Veterans in work rehabilitation. We have also combined our validated social cognitive training program with oxytocin (OT), which is believed to enhance the salience of social cues, and showed a beneficial effect on some aspects of social cognition. We recently demonstrated that OT increased mu-suppression as measured by EEG in response to social stimuli, providing dosing guidance for future studies. MIRECC investigators have two ongoing OT studies. One tests whether administration of intranasal OT just before social cognitive training facilitates learning of higher level mentalizing skills, including empathy. Another evaluates whether twice daily OT administration improves social functioning and negative symptoms and whether hypo-activation in the right temporo-parietal junction during theory of mind tasks correlates with negative symptoms in Veterans with schizophrenia. These treatment studies have direct implications for care of Veterans with schizophrenia.
MIRECC investigators are also conducting translational preclinical studies in rodent models to understand social deficits and consequences of social isolation, using cross-species tasks similar to tasks in humans. Using a neurodevelopmental model relevant to schizophrenia in which mice are exposed to an immune challenge in utero, we showed that social deficits can be attenuated with pharmacological interventions (e.g., purinergic or 5-HT2A antagonist) and that social deficits in the model are correlated with reversal learning deficits and associated with frontal cortex gene expression. Dr. Powell is currently investigating the neural circuitry of these deficits using state of the art optogenetic techniques. She’s also examining the impact of adolescent/early adulthood social isolation in rodents on cognitive deficits and the corresponding neural circuitry and pharmacological treatments to reverse them. These studies will form the foundation for later treatment interventions for Veterans with schizophrenia.
Service Use and Contextual Factors
(e.g., poverty, housing status, systemic racism, and gender disparities)
Veteran behaviors (e.g., use of mental health care), as well as community factors, policies, and the organization of services (i.e., context) are critical determinants of clinical and functional outcomes for Veterans with SMI. Our Health Services Implementation Unit has a long trajectory of identifying service use disparities for Veterans with SMI and developing strategies to address these disparities. More recently, we have focused on the urgent, national VA priority to end Veteran homelessness. Veterans who remain homeless despite VA’s tremendous investment in health and housing services are extraordinarily vulnerable. We have developed, adapted, and tested evidence-based practices for Veterans with SMI in the setting and context of VA homeless programs, including a group intervention that improves executive functioning (PI: Greenberg), social skills training (PI: Gabrielian), and Intensive Housing First, which engages high-need consumers in assertive community services (PI: Young). We also aimed to identify effective practices (e.g., implementation facilitation) that support the scale up, spread, and sustainment of EBPs across the broad range of programs that serve homeless Veterans. The QUERI-funded implementation initiatives include Bridge QUERI which implemented a psychosocial intervention for homeless Veterans with co-occurring disorders (PI: Smelson, Site PI: Gabrielian) and a QUERI Partnered Implementation Initiative which implemented Critical Time Intervention for Veterans engaged in one of VA’s homeless programs. Interactions between the MIRECC and THRIVe – the recently funded RR&D Center on community integration for recently homeless Veterans – have allowed us to study other contextual factors, including the effects of neighborhood characteristics (e.g., socioeconomic status, racial/ethnic diversity) and housing type (e.g., congregate versus community-based settings) on functioning among Veterans with homeless experiences.
Aligned with the national VA priority to improve women Veterans’ health, Dr. Goodsmith is studying VA health service engagement among women Veterans with SMI. This project expands the scope of our research to encompass gender disparities in this vulnerable population.
Education Research Programs
The MIRECC’s Education and Dissemination Unit (EDU) conducts research to identify education needs of Veterans, caretakers, clinicians, and community providers. Here are a few areas of research led by the EDU.
Mental Health Information Seeking Practices of Veterans
Understanding individuals’ health information-seeking practices is a necessary precursor for effective delivery of health information. However, little is known about how individuals with mental health difficulties access information about their health. Therefore, the EDU interviewed 81 Veterans, primarily with SMI, to explore their health information-seeking practices. The study aims to understand their needs and preferences regarding the content, source, and modality of the health information they seek and how individual characteristics (e.g., age, sex, technology access, mental health diagnosis, and cognition) influence their information seeking practices. The study will inform the development and dissemination of future education products created by our MIRECC.
Dissemination of Veteran-Centered Brief Family Consultation
The EDU responded to a request from VA Family Services to develop education materials on Veteran-Centered Brief Family Consultation. Dr. Niv developed a five-module web-based training for clinicians as well as an instructional set. The EDU has completed data collection for a randomized controlled trial comparing web-based training of family consultation to in-person training to a control group which has not received any training. The study, conducted at five sites in VISN 22, found that web-based training and in-person training both led to improved knowledge of the intervention and self-efficacy in administering it compared to the control group. Outcomes in the two active training conditions were similar. Fidelity to the intervention and implementation will also be examined.
Meeting the Needs of Student Veterans
To enhance care in the community, the EDU completed a needs assessment of 93 Veteran Coordinators and 80 directors of college counseling centers. The study aimed to identify the role Veteran Coordinators play in Veterans’ mental health, the mental health services available to student Veterans on college campuses, the mental health education needs of Veteran Coordinators and clinical staff to better support student Veterans, and what types of education they would prefer.
Residential Weight Intervention for SMI
Lack of control over the foods purchased and prepared has been identified as a major obstacle to weight loss among SMI adults. Innovative approaches to address such barriers are needed to help improve weight loss efforts targeting mentally ill consumers. One such approach is to develop weight loss interventions targeting residential settings in which individuals with SMI reside. Dr. Niv conducted a study which applies the ANGELO process, a framework for identifying and prioritizing environmental interventions for obesity, to development of a residential-based weight loss intervention. The EDU conducted a situational analysis with 71 residential facilities that house Veterans with SMI to 1) identify current practices regarding food service and wellness, determinants of current practices, variations from federal nutrition and physical activity guidelines, staff knowledge, readiness to change, and barriers and facilitators to improvement, 2) identify and prioritize potential behaviors to target, knowledge and skill gaps to address, and environmental barriers to overcome with the input of stakeholders, and 3) develop an action plan (i.e., specific objectives and implementation strategies to achieve each objective). The EDU is now developing education materials based on the findings of the study.



















